CHAPTER – 1 “Matter in our Surroundings”
conceptdetails
KEY CONCEPTS : [ *rating as per the significance of concept]
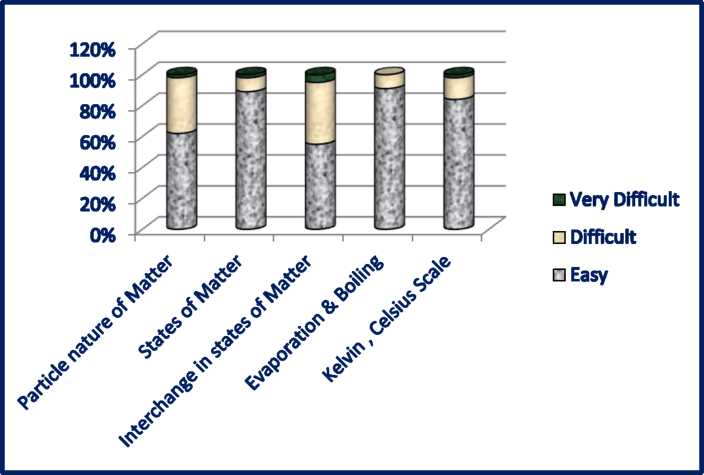
1. Particle nature of Matter | *** |
2. States of Matter | **** |
3. Interchange in states of Matter | ***** |
4. Evaporation & Boiling | **** |
5. Kelvin , Celsius scale | *** |
-I- Pre requisites
- Definition of matter.
- Elementary idea of three physical states of matter.
SURVEY ANALYSIS
Conceptual levels of comprehension on the basis of feedback taken from the students
1. Particle Nature of Matter j
[ refer NCERT text book activities 1.1 to 1.8 1
- Anything that occupies space and has mass and is felt by senses is called matter.
- Matter is the form of five basic elements the Panch tatva – air , earth fire , sky and water.
- Characteristics of particles of matter
- Made of tiny particles.
- Vacant spaces exist in particles.
- Particles are in continuous motion.
- Particles are held together by forces of attraction.
Q.1 Define matter.
Q.2 What happens if you put copper sulphate crystals in water?
[ refer NCERT text book activities 1.9 to 1.111
Basis of Classification of Types
- Based upon particle arrangement
- Based upon energy of particles
- Based upon distance between particles
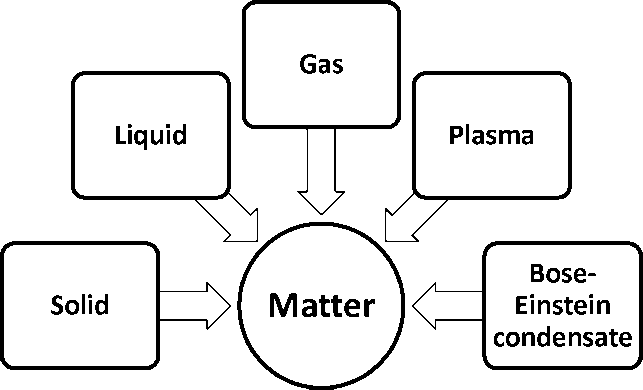
- Five states of matter


- Fixed shape and definite volume .
- Not fixed shape but fixed volume.
- Neither fixed shape nor fixed volume.
- Inter particle distances are smallest.
- Inter particle distances are larger.
- Inter particle distances are largest.
Almost incompressible.
Incompressible.
Highly compressible.
- High density and do not diffuse.
- Inter particle forces of attraction are strongest.
- Constituent particles are very closely packed.
- Density is lower than solids and diffuse.
- Inter particle forces of attraction are weaker than solids .
- Constituent particles are less closely packed.
- Density is least and diffuse.
- Inter particle forces of attraction are weakest.
- Constituent particles are free to move about.

A plasma is an ionized gas.
A plasma is a very good conductor of electricity and is affected by magnetic fields.
Plasma, like gases have an indefinite shape and an indefinite volume. Ex. Ionized gas
- A BEC is a state of matter that can arise at very low temperatures.

- The scientists who worked with the Bose-Einstein condensate received a Nobel Prize for their work in 1995.
- The BEC is all about molecules that are really close to each other (even closer than atoms in a solid).
Microscopic Explanation for Properties of Solids
Solids have a definite shape and a definite volume because the particles are locked into place | Solids do not flow easily because the particles cannot move/slide past one another past one another | Solids are not easily compressible because there is little free space between particles | |
Microscopic Explanation for Properties of Liauids | |||
Liquids are not easily compressible and have a definite volume because there is little free space between particles. | Liquids have an indefinite shape because the particles can lide past one another. past one another. | Liquids flow easily because the particles can move/slide past one another. | |
Microscopic Explanation for Properties of Gases | |||
Gases are easily compressible because there is a great deal of free space between particles | Gases have an indefinite shape and an indefinite volume because the particles can move past one another. | ||
Gases flow very easily because the particles randomly move past one another. | |||
(non -evaluative)^ | |||
Microscopic Explanation for Properties of Plasmas | |||
Plasmas have an indefinite shape and an indefinite volume because the particles can move past one another. | Plasmas are easily compressible because there is a great deal of free space between particles. | Plasmas are good conductors of electricity &are affected by magnetic fields because they are composed of ions | |
Microscopic Explanation for Properties of BEC | |||
Particles are less energetic than solids because Exist at very low temperature . | Particles are literally indistinguishable because they are locked into same space . | BEC shows superfluidity because Particles can flow without friction. | |
Q.1 A substance has a definite volume but no definite shape ? State whether this substance is a solid, a liquid or a gas.
Q.2 Arrange the following substances in increasing order of force of attraction between the particles. (a) Milk (b) Salt (c) Oxygen.
Q.3 A substance has neither a fixed shape nor a fixed volume . State whether it is a solid, a liquid or a gas.
Q.4 The melting point of a substance is below the room temperature . Predict its physical state.
3.Interchange in states of matter
[ refer NCERT text book activities 1.12 to 1.14 1 Matter Can Change its State
Water can exist in three states of matter –
- Solid, as ice ,
- Liquid, as the familiar water, and
- Gas, as water vapour.
Sublimation : The changing of solid directly into vapours on heating & vapours into solid on cooling. Ex. Ammonium chloride , camphor & iodine.
- Effect of change in temperature
The temperature effect on heating a solid varies depending on the nature of the solid & the conditions required in bringing the change .
- On increasing the temperature of solids, the kinetic energy of the particles increases which overcomes the forces of attraction between the particles thereby solid melts and is converted to a liquid.
- The temperature at which a solid melts to become a liquid at the atmospheric pressure is called its melting point.
- The melting point of ice is 273.16 K.
- The process of melting, that is, change of solid state into liquid state is also known as fusion.
- Effect of Change of Pressure
- Increasing or decreasing the pressure can change the state of matter. Applying pressure and reducing temperature can liquefy gases.
- Solid carbon dioxide (CO2) is stored under high pressure. Solid CO2 gets converted directly to gaseous state on decrease of pressure to 1 atmosphere without coming into liquid state. This is the reason that solid carbon dioxide is also known as dry ice. Latent Heat :
The hidden heat which breaks the force of attraction between the molecules during
change of state.
Fusion | Vaporisation |
Heat energy required to change 1kg of solid into liquid. | Heat energy required to change 1kg of liquid to gas at atmospheric pressure at its boiling point. |
Thus, we can say that pressure and temperature determine the state of a substance, whether it will be solid, liquid or gas.
[ refer fig. 1.9 NCERT Text Book, page-8 ]
Q.1 | What is vapour ? |
Q.2 Name the temperature at which the solid and liquid states of substance can exist | |
together . | |
Q.3 | What is the effect of pressure on boiling point? |
Q.4 | Name any two substances which sublime. |
Q.5 | Define Condensation. |
Q.6 For any substance, why does the temperature remain constant during the | |
change of state? | |
- Particles of matter are always moving and are never at rest.
- At a given temperature in any gas, liquid or solid, there are particles with different
amounts of kinetic energy.
- In the case of liquids, a small fraction of particles at the surface, having higher kinetic energy, is able to break away from the forces of attraction of other particles and gets converted into vapour .
- This phenomenon of change of a liquid into vapours at any temperature below its boiling point is called evaporation.
- Factors Affecting Evaporation
- The rate of evaporation increases with an increase of surface area.
- With the increase of temperature, more number of particles get enough kinetic energy
to go into the vapour state.
- Humidity is the amount of water vapour present in air. The air around us cannot hold more than a definite amount of water vapour at a given temperature. If the amount of water in air is already high, the rate of evaporation decreases.
- Wind speed : the higher the wind speed , the more evaporation.
The particles of liquid absorb energy from the surrounding to regain the energy lost during evaporation,
- Boiling is a bulk phenomenon. Particles from the bulk (whole) of the liquid change into vapour state.
- Evaporation is a surface phenomenon. Particles from the surface gain enough energy to overcome the forces of attraction present in the liquid and change into the vapour state.
Q.1 Which is the slow process, Evaporation or Boiling ?
Q.2 State the effect of surface area on rate of evaporation.
Q.3 Why are we able to sip hot tea faster from saucer rather than from a cup?
- Kelvin is the SI unit of temperature, 00 C =273.16 K. we take 00 C = 273 K.
- SI unit of temperature is Kelvin. T (K)= T (oC) +273
- Kelvin scale of temperature has always positive sign , hence regarded as better scale than Celsius.
- Atmosphere (atm) is a unit of measuring pressure exerted by a gas. The SI unit of pressure is Pascal (Pa):
- 1 atmosphere = 1.01 x (10 to the power 5) Pa. The pressure of air in atmosphere is called
atmospheric pressure. The atmospheric pressure at sea level is 1 atmosphere, and is
taken as the normal atmospheric pressure.
Q.1 What is the SI unit of temperature?
Q.2 Kelvin scale of temperature is regarded as better scale than Celsius. Why?
Q.3 Convert 10oC into Kelvin scale.
- Mark Questions:
- Pressure on the surface of a gas is increased. What will happen to the inter particle forces?
- Name the three states of matter.
- What happens when a liquid is heated ?
- A gas can exert pressure on the walls of the container. Assign reason.
- Convert the following temperature to Kelvin Scale (a) 100°C (b) 37°C
- What is meant by density?
- Give the characteristics of the particles of matter.
- Water droplets seen on the outer surface of a glass containing ice-cold water is due
to .
- Change of gaseous state directly to solid state without going through liquid sate is
called .
- . is a surface phenomenon.
- Marks Questions:
- Define Latent heat of vaporisation.
- Explain why temperature remain constant during the change of state of any substance?
- Define Sublimation with examples.
- *Do we sweat more on a dry day or humid day ? Justify your reason.
- Why do we see water droplets on the outer surface of a glass containing ice cold water?
- Convert the following temperature to the Kelvin scale (a) 25°C (b) 373°C
- List two properties that liquids have in common with solids.
- List two properties that liquids have in common with gases.
- *What will happen to the melting point temperature of ice if some common salt is added to it? Justify your answer.
- *How will you show that air has maximum compressibility?
- Marks Questions:
- Define the term (a) Latent heat of fusion (b) Latent heat of vaporization
- *State the effect of (i) surface area (ii) nature of the liquid on the rate of evaporation.
- *Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Why?
- What is the physical state of water at 250°C, 100°C, 0°C?
- Give reasons :
- A sponge can be pressed easily; still it is called a solid.
- Water vapours have more energy than water at same temperature.
6 . What are intermolecular forces ? How are these related to the three states of matter ?
- Is it possible to liquify atmospheric gases? If yes, suggest a method.
1. a) What is meant by evaporation? What are the factors on which the rate of evaporation depend upon?
- How does evaporation causes cooling?
- State the properties of all the five states of matter.
- Define : Melting point , Freezing point & Boiling point
- Particle nature of matter.
- All five states of matter & their behaviour
- Inter conversion of states of matter
- Latent heat
- Conversion between Kelvin scale & Celsius scale