CBSE Class 12 Chemistry
Quick Revision Notes
Chapter 5
Surface Chemistry
- The accumulation of molecular species at the surface rather than in the bulk of a solid or liquid is termed as adsorption.
- It is a surface phenomenon.
- The concentration of adsorbate increases only at the surface of the adsorbent.
- Adsorbate: It is the substance which is being adsorbed on the surface of another substance.
- Adsorbent: It is the substance present in bulk, on the surface of which adsorption is taking place.
- Desorption: It is the process of removing an adsorbed substance from a surface on which it is adsorbed.
- Absorption:
- It is the phenomenon in which a substance is uniformly distributed throughout the bulk of the solid.
- It is a bulk phenomenon.
- The concentration is uniform throughout the bulk of solid.
- Sorption: When adsorption and absorption take place simultaneously, it is called sorption.
- Enthalpy or heat of adsorption: Since, adsorption occurs with release in energy, i.e., it is exothermic in nature. The enthalpy change for the adsorption of one mole of an adsorbate on the surface of adsorbent is called enthalpy or heat of adsorption.
- Types of adsorption: There are different types of adsorption namely,
- Physical adsorption
- Chemical adsorption
- If the adsorbate is held on a surface of adsorbent by weak van der Waals’ forces, the adsorption is called physical adsorption or physisorption.
- It is non-specific.
- It is reversible.
- The amount of gas depends upon nature of gas, i.e., easily liquefiable gases like NH3, CO2, gas adsorbed to greater extent than H2 and He. Higher the critical
temperature of gas, more will be the extent of adsorption.
- The extent of adsorption increases with increase in surface area, e.g. porous and finely divided metals are good adsorbents.
- There are weak van der Waals’ forces of attraction between adsorbate and adsorbent.
- It has low enthalpy of adsorption (20 – 40 kJ mol-1).
- Low temperature is favourable.
- No appreciable activation energy is needed.
- It forms multimolecular layers.
- If the forces holding the adsorbate are as strong as in chemical bonds, the adsorption process is known as chemical adsorption of chemisorption.
- It is highly specific.
- It is irreversible.
- The amount of gas adsorbed is not related to critical temperature of the gas.
- It also increases with increase in surface area.
- There is strong force of attraction similar to chemical bond.
- It has enthalpy heat of adsorption (180 – 240 kJ mol-1).
- High temperature is favourable.
- High activation energy is sometimes needed.
- It forms unimolecular layers.
a. Nature of adsorbate: Physical adsorption is non-specific in nature and therefore every gas gets adsorbed on the surface of any solid to a lesser or greater extent. However, easily liquefiable gases like NH3,HCl, CO2, etc. which have higher critical temperatures are
absorbed to greater extent whereas H2, O2, N2 etc. are adsorbed to lesser extent. The
chemical adsorption being highly specific, therefore, a gas gets adsorbed on specific solid only if it enters into chemical combination with it.
- Nature of adsorbent: Activated carbon, metal oxides like aluminum oxide, silica gel and clay are commonly used adsorbents. They have their specific adsorption properties depending upon pores.
- Specific area of the adsorbent: The greater the specific area, more will be the extent of adsorption. That is why porous or finely divided forms of adsorbents adsorb larger quantities of adsorbate. The pores should be large enough to allow the gas molecules to enter.
- Pressure of the gas: Physical adsorption increases with increase in pressure.
- Adsorption isotherm:
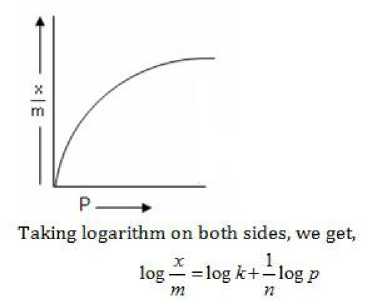
- The variation in the amount of gas adsorbed by the adsorbent with pressure at constant temperature can be expressed by means of a curve is termed as adsorption isotherm.
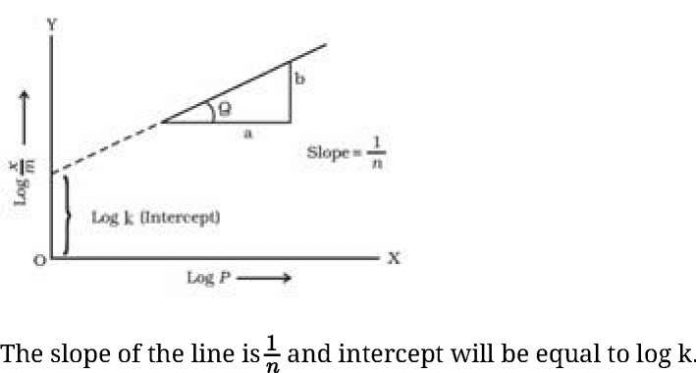
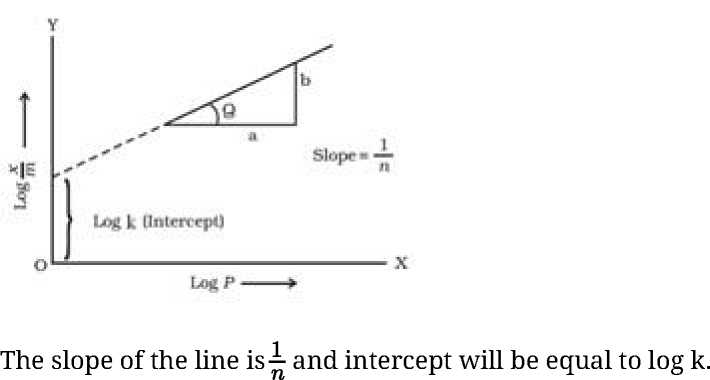
- Freundlich Adsorption isotherm: The relationship between ^ and pressure of the gas at constant temperature is called adsorption isotherm and is given by
Where x- mass of the gas adsorbed on mass m of the adsorbent and the gas at a particular temperature k and n depends upon the nature of gas
- The solid^-first increases with increase in pressure at low pressure but becomes independent of pressure at high pressure.
Taking logarithm on both sides, we get,
Iog^ = IogA:+ ^logp
• If we plot a graph between log^ and log P, we get a straight line.
- Catalyst: These are substances which alter the rate of a chemical reaction and themselves remain chemically and quantitatively unchanged after the reactionand the phenomenon is known as catalysis.
- Promoters: These are the substances which increase the activity of catalyst. Example –
Mo is promoter whereas Fe is catalyst in Haber’s Process.
Fe(s)/Mo(s)
N2{g) + 3#2(s) ► 2NH3{g)
- Catalytic poisons (Inhibitors): These are the substances which decrease the activity of catalyst. Example -Arsenic acts as catalytic poison in the manufacture of sulphuric acid by ‘contact process.’
- Types of catalysis:
There are two types of catalysis namely,
- Homogeneous catalysis: When the catalyst and the reactants are in the same phase, this kind of catalytic process is known as homogeneous catalysis.
- Heterogeneous catalysis: When the catalyst and the reactants are in different phases, the catalytic process is said to be heterogeneous catalysis.
- Activity of catalyst: It is the ability of a catalyst to increase the rate of a chemical reaction.
- Selectivity of catalyst: It is the ability of catalyst to direct a reaction to yield a particular
product (excluding others).
For example: CO and H2 react to form different products in presence of different catalysts as follows:
Ni
1.
Cu/ZnO-Cr2 O3
2.
Cu
3.
- Shape – selective catalysis: It is the catalysis which depends upon the pore structure of the catalyst and molecular size of reactant and product molecules. Example – Zeolites are shape – selective catalysts due to their honey- comb structure.
- Enzymes: These are complex nitrogenous organic compounds which are produced by living plants and animals. They are actually protein molecules of high molecular mass. They are biochemical catalysts
- Steps of enzyme catalysis:
- Binding of enzyme to substrate to form an activated complex.
- Decomposition of the activated complex to form product.
- They are highly efficient. One molecule of an enzyme can transform 106 molecules of reactants per minute.
- They are highly specific in nature. Example – Urease catalysis hydrolysis of urea only.
- They are active at optimum temperature (298 – 310 K). The rate of enzyme catalysed reaction becomes maximum at a definite temperature called the optimum temperature.
- They are highly active at a specific pH called optimum pH.
- Enzymatic activity can be increased in presence of coenzymes which can be called as promoters.
1 2 j. 2 4-
- Activators are generally metal ions Na , Co2 and Cu2 etc. They weakly bind to enzyme and increase its activity.
- Influence of inhibitors (poison): Enzymes can also be inhibited or poisoned by the presence of certain substances.
- It is homogeneous.
- The diameter of the particles is less than 1 nm.
- It passes through filter paper.
- Its particles cannot be seen under a microscope.
- It appears to be homogeneous but is actually heterogeneous.
- The diameter of the particles is 1 nm to 1000 nm.
- It passes through ordinary filter paper but not through ultra-filters.
- Its particles can be seen by a powerful microscope due to scattering of light.
- It is heterogeneous.
- The diameter of the particles are larger than 1000 nm.
- It does not pass through filter paper.
- Its particles can be seen even with naked eye.
- Dispersed phase: It is the substance which is dispersed as very fine particles.
- Dispersion medium: It is the substance present in larger quantity.
- Classification of colloids on the basis of the physical state of dispersed phase and dispersion medium:
|
Name |
Dispersed phase |
Dispersed medium |
Examples |
|
Solid sol |
solid |
Solid |
Coloured gem stones |
|
Sol |
Solid |
Liquid |
Paints |
|
Aerosol |
Solid |
Gas |
Smoke, dust |
|
Gel |
Liquid |
Solid |
Cheese, jellies |
|
Emulsion |
Liquid |
Liquid |
Hair cream, milk |
|
Aerosol |
Liquid |
Gas |
Mist, fog, cloud |
|
Solid sol |
Gas |
Solid |
Foam rubber, pumice stone |
|
Foam |
Gas |
Liquid |
Whipped cream |
- Classification of colloids on the basis of nature of interaction between dispersed phase and dispersion medium, the colloids are classified into two types namely,
- Lyophobic sols
- Lyophilic sols
- These colloids are liquid hating.
- In these colloids the particles of dispersed phase have no affinity for the dispersion medium.
- They are not stable.
- They can be prepared by mixing substances directly.
- They need stabilizing agents for their preservation.
- They are irreversible sols.
- These colloids are liquid loving.
- In these colloids, the particles of dispersed phase have great affinity for the dispersion medium.
- They are stable.
- They cannot be prepared by mixing substances directly. They are prepared only by
special methods.
- They do not need stabilizing agents for their preservation.
- They are reversible sols.
- Classification of colloids on the basis of types of particles of the dispersed phase:
There are three types of colloids based on the type of dispersed phase, namely,
- Multimolecular colloids: The colloids in which the colloidal particles consist of aggregates of atoms or small molecules. The diameter of the colloidal particle formed is less than 1 nm.
- Macromolecular colloids: These are the colloids in which the dispersed particles are themselves large molecules (usually polymers). Since these molecules have dimensions comparable to those of colloids particles, their dispersions are called macromolecular colloids, e.g., proteins, starch and cellulose form macromolecular colloids.
- Associated colloids (Micelles): Those colloids which behave as normal, strong electrolytes at low concentrations, but show colloidal properties at higherconcentrations due to the formation of aggregated particles of colloidal dimensions. Such substances are also referred to as associated colloids.
- Kraft Temperature (Tk):Micelles are formed only above a certain temperature called Kraft temperature.
- Critical Micelle Concentration (CMC): Micelles are formed only above a particular concentration called critical micelle concentration.
- Soaps: These are are sodium or potassium salts of higher fatty acids e.g., sodium stearate CH3(CH2)i6COO-Na+
- Methods of preparation of colloids:
- Chemical methods: Colloids can be prepared by chemical reactions leading to the formation of molecules. These molecules aggregate leading to formation of sols.
- Electrical disintegration or Bredig’s Arc method: In this method, electric arc is struck
between electrodes of the metal immersed in the dispersion medium. The intense heat produced vaporizes the metal which then condenses to form particles of colloidal size.
- Peptization: It is the process of converting a precipitate into colloidal sol by shaking it with dispersion medium in the presence of a small amount of electrolyte. The electrolyte used for this purpose is called peptizing agent.
- Dialysis: It is a process of removing a dissolved substance from a colloidal solution by means of diffusion through a suitable membrane.
- Electro dialysis. The process of dialysis is quite slow. It can be made faster by applying an electric field if the dissolved substance in the impure colloidal solution is only an electrolyte.
- Ultrafiltration: It is the process of separating the colloidal particles from the solvent and soluble solutes present in the colloidal solution by specially prepared filters, which are permeable to all substances except the colloidal particles.
- Ultracentrifugation: In this process, the colloidal solution is taken in a tube which is placed in ultracentrifuge. On rotating the tube at very high speed, the colloidal particles settle down at the bottom of the tube and the impurities remain in solution. The settled particles are mixed with dispersion medium to regenerate the sol.
Positively charged colloidal particles:
- These include hydrated metallic oxides such as Fe2O3.#H2O, Cr2O3.a;H2O, Al2O3.
CCH2O
- Basic dye stuff like malachite green, methylene blue sols.
- Example – Haemoglobin (blood).
Negatively charged colloidal particles:
- Metallic sulphides like As2S3, Sb2S3 sols.
- Acid dye stuff like eosin, methyl orange, Congo red sols.
- Examples – Starch sol, gum, gelatin, clay, charcoal, egg albumin, etc.
- Colour: The colour of colloidal solution depends upon the wavelength of light scattered by the colloidal particles which in turn depends upon the nature and size of particles. The colour also depends upon the manner in which light is received by the observer.
- Brownian movement: Colloidal particles move in zig – zag path. This type of motion is due to colliding molecules of dispersion medium constantly with colloidal particles.
- Colligative properties: The values of colligative properties (osmotic pressure, lowering in vapour pressure, depression in freezing point and elevation in boiling point) are of small order as compared to values shown by true solutions at the same concentrations.
- Tyndall effect: The scattering of a beam of light by colloidal particles is called Tyndall effect. The bright cone of light is called the Tyndall cone.
- Charge on colloidal particles: Colloidal particles always carry an electric charge. The nature of this charge is the same on all the particles in a given colloidal solution and may be either positive or negative.
- Helmholtz electrical double layer: When the colloidal particles acquire negative or positive charge by selective adsorption of one of the ions, it attracts counter ions from the medium forming a second layer. The combination of these two layers of opposite charges around colloidal particles is called Helmholtz electrical double layer.
- Electrokinetic potential or zeta potential: The potential difference between the fixed layer and the diffused layer of opposite charges is called electrokinetic potential or zeta potential.
- Electrophoresis: The movement of colloidal particles under an applied electric potential is called electrophoresis.
- Coagulation or precipitation: The process of settling of colloidal particles as precipitate is called coagulation.
• Hardy – Schulze rules:
- Oppositely charged ions are effective for coagulation.
- The coagulating power of electrolyte increases with increase in charge on the ions used for coagulation. Examples – Al3+> Ba2+> Na+ for negatively charged colloids. Fe (CN)6]4->
PO^ >S0^ >Cl– for positively charged colloids.
- Types of emulsions:
- Water dispersed in oil: When water is the dispersed phase and oil is the dispersion medium. E.g. butter
- Oil dispersed in water: When oil is the dispersed phase and water is the dispersion medium. E.g. milk
- Emulsification: It is the process of stabilizing an emulsion by means of an emulsifier.
- Emulsifying agent or emulsifier: These are the substances which are added to stabilize the emulsions. Examples – soaps, gum
- Demulsification: It is the process of breaking an emulsion into its constituent liquidsby freezing, boiling, centrifugation or some chemical methods.