CBSE Class 12 Chemistry
Quick Revision Notes
Chapter 11
Alcohols, Phenols and Ethers
MtQianol
(Akahd)
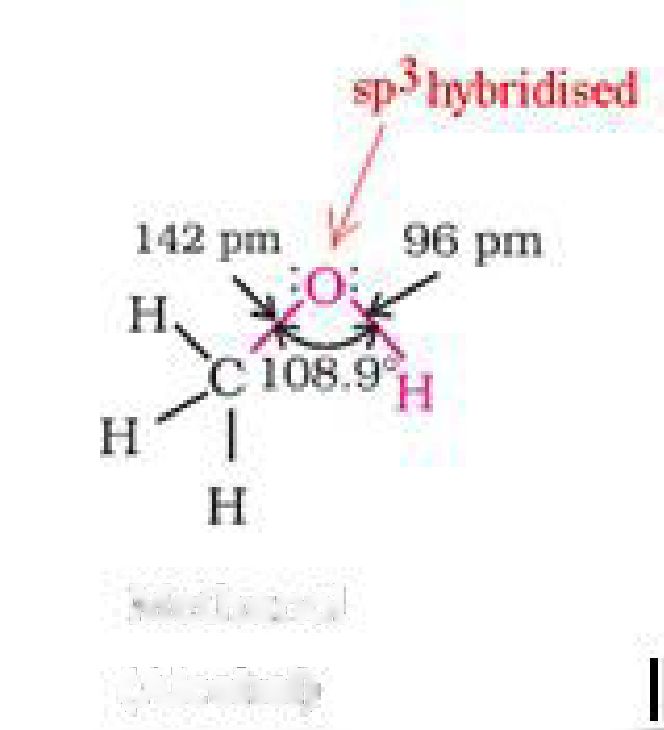
• Structure of alcohols:
Preparation of alcohols:
- From alkene
Acid catalysed hydration
(H 20,H+)
Mark, Addition
Or
Hydroboration – oxidation
B2H^H2O2IOH-
Mark, addition
Product is anti mark
Alkene > Alcohol
- From esters
H2 catalyst
Esters > Alcohol
- From aldehydes and ketones
H2jPdor
NaBHi OrLiAlHi OrGrignard’s reagent
Aldehyde and ketone
- From carboxylic acids
LiAlHi H2O
Alcoho 1 <r
Alcoho 1 i Carboxylic acids
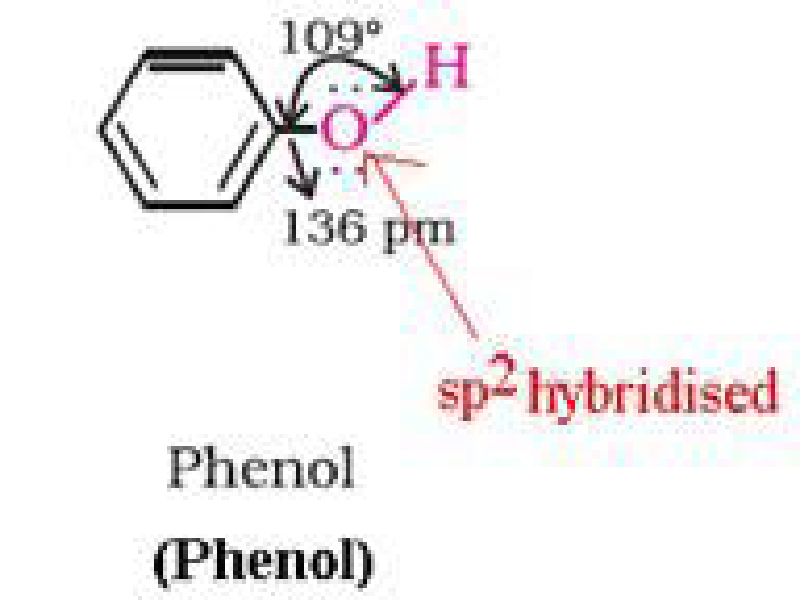
• Structure of phenols:
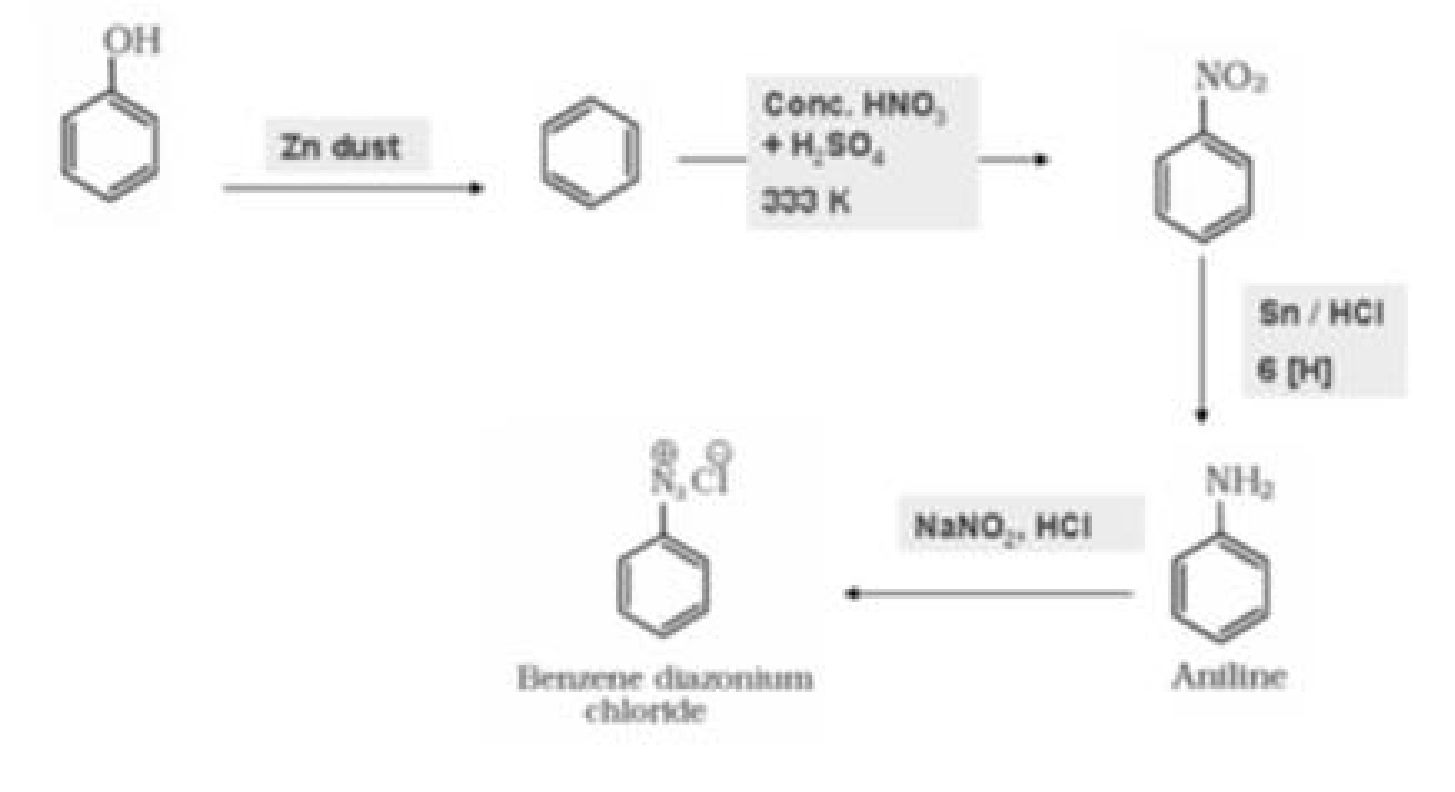
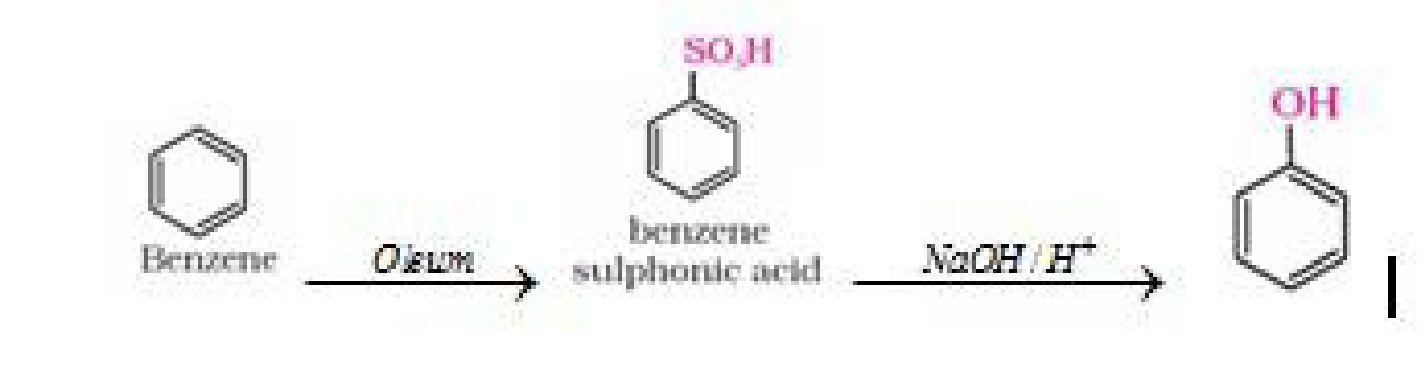
a) From benzene
b) From chlorobenzene

c) From cumene
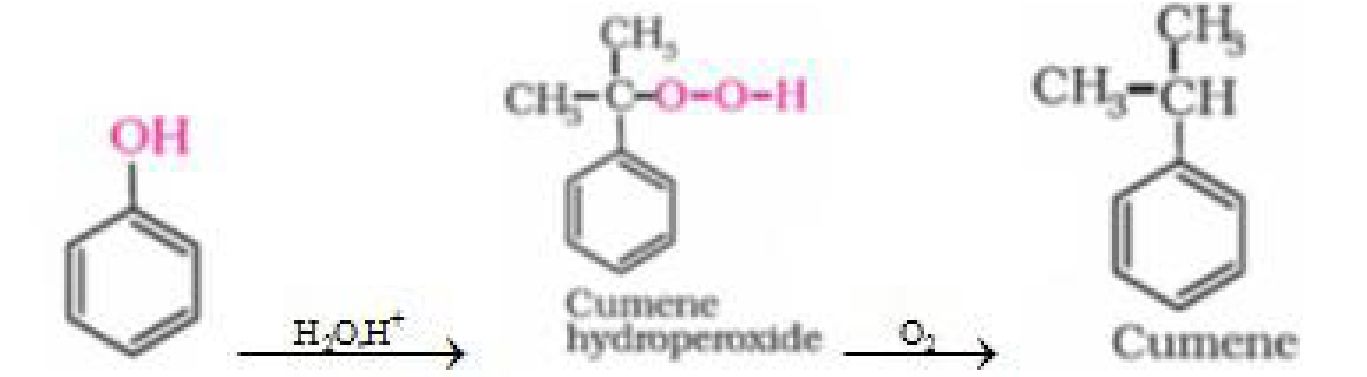
d) From aniline
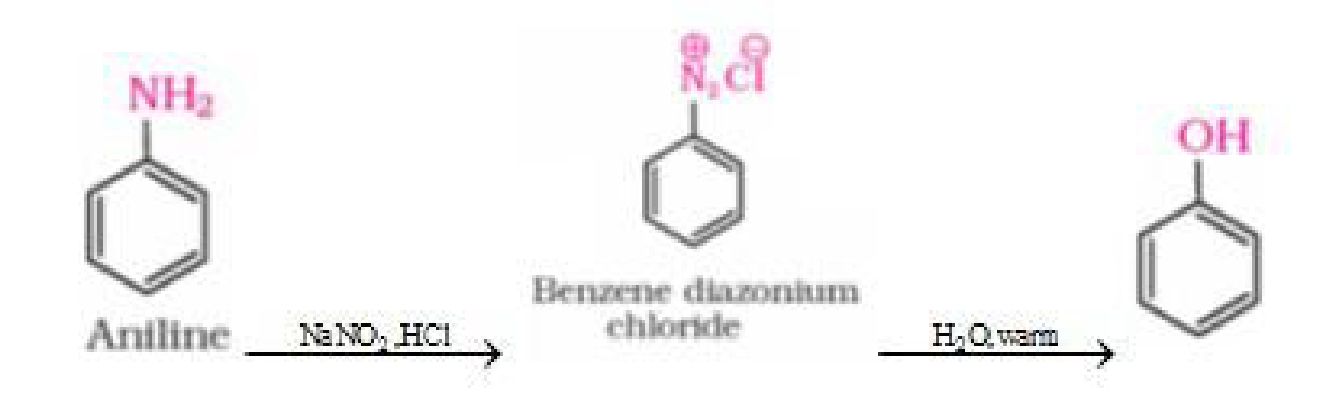
Physical properties of alcohols and phenols:
- Boiling points: Boiling points of alcohols and phenols are higher in comparison to other
classes of compounds. This is because the -OH group in alcohols and phenols is involved in
intermolecular hydrogen bonding.
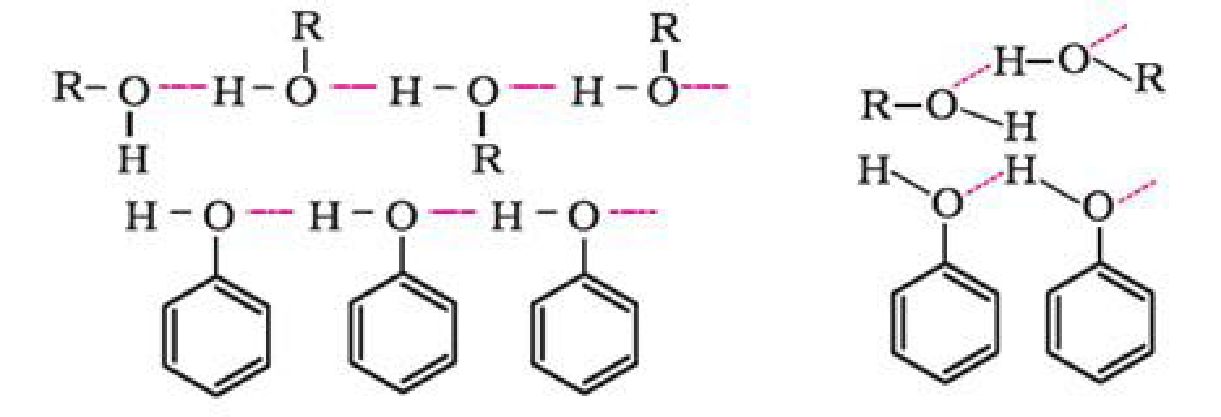
The boiling points of alcohols and phenols increase with increase in the number of carbon
atoms. This is because of increase in van der Waals forces with increase in surface area.
In alcohols, the boiling points decrease with increase of branching in carbon chain. This is
because of decrease in van der Waals forces with decrease in surface area.
- Solubility: Solubility of alcohols and phenols are soluble in water due to their ability to
form hydrogen bonds with water molecules. The solubility of alcohols decreases with
increase in size of alkyl/aryl (hydrophobic) groups.
• Chemical properties of alcohols:
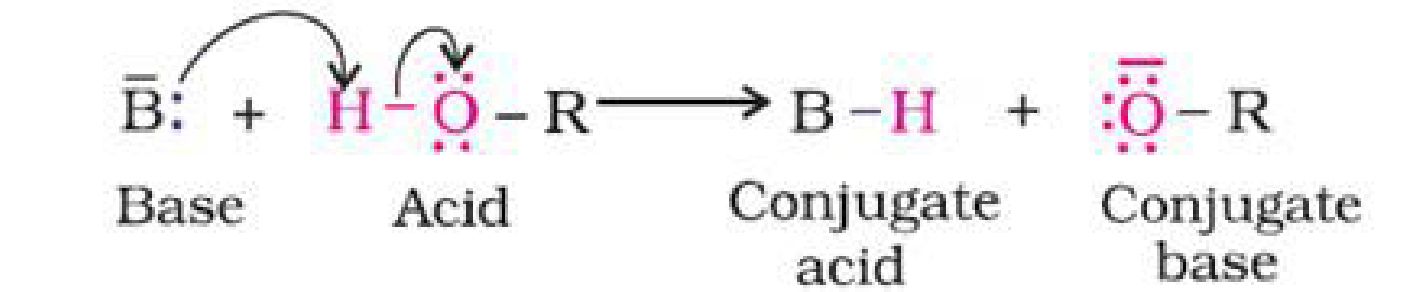
- Reactions involving cleavage of O-H bond: Alcohols react as nucleophiles:
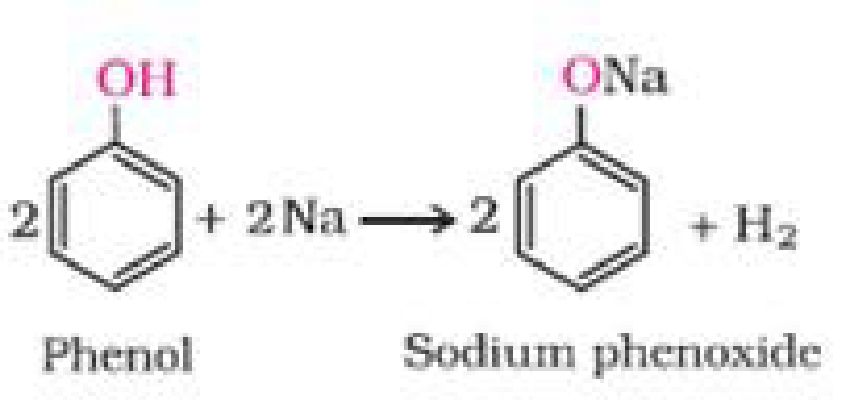
- Reaction with metals
2 R – O – H + 2 Na ^ 2 R – O – Na + H2
Sodium alkoxide
- Esterification reaction
RO – H + R’ – COOH S ROCOR’ + H2 O
Alcohol
RO – H + (R!C02)0%- ROCOR + RCOOH
Alcohol
Pnridinp
RO – H + R!COCl — ► R – OCORf + HCl
Alcohol
- Reactions of alcohols involving cleavage of carbon – oxygen (C-O) bond:
- Reaction with hydrogen halides
c0nc.HCl+ZnCl2/Lucas reagent
- Reaction with phosphorus trihalides
3ROH + PX3 ^ 3R – X + H3P03(X = Cl, Br)
- Dehydration reaction
P10tic&cids(conc.H2SO40rHzPO4)OrCatalysts(anhyd.ZnCl2 or alumina)
- . Oxidation reaction
Acidified potassium permanganat
CU,573k
Or
CiOs
Or
PCC
CU,573k
Or
CrO3
ii)
CU,573k
Or
KMnOi
iii)
• Chemical properties of phenols:
I. Reactions involving cleavage of O-H bond: Alcohols react as nucleophiles:
a) Reaction with metals
b) Esterification reaction
Ar – OH + H – COOH ^> Ar – OCOR + H2O
Phenol
Ar – OH + (RCO)2O «■ Ar – OCOR + RCOOH
Phenol
P? m*n m 77 p
Ar – OiJ + RCOCl — > ArOCOR1 + iJC7
Phenol
II. Other chemical reactions of phenols:
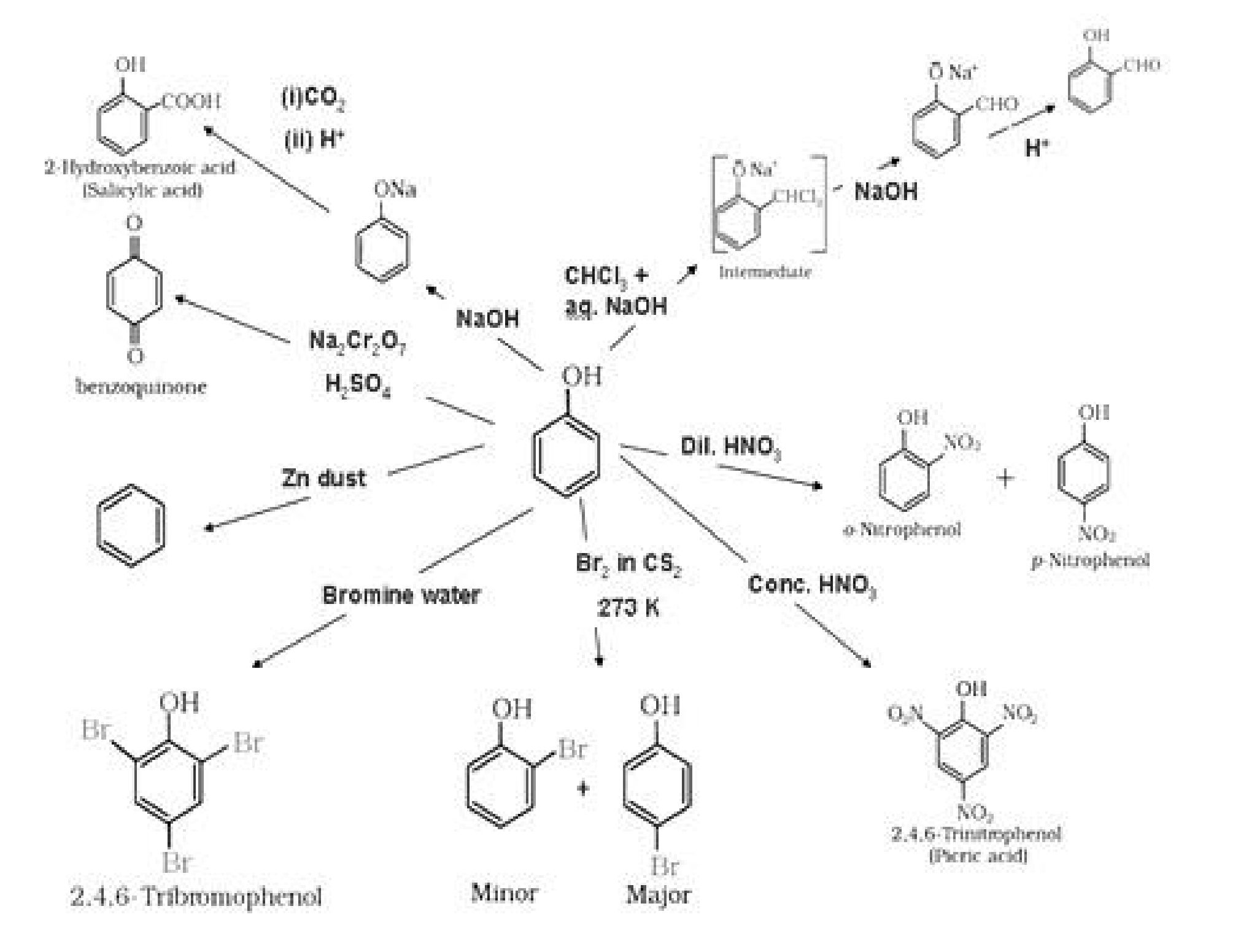
- Acidic nature of phenol and alcohol:
a). Phenol > H2O > Primary alcohol > Secondary alcohol > Tertiary alcohol.
The acidic character of alcohols is due to the polar nature of O-H bond. Alkyl group is an
electron-releasing group (-CH3, -C2H5) or it has electron releasing inductive effect (+I effect).
Due to +I effect of alkyl groups, the electron density on oxygen increases. This decreases the
/10
Material Downloaded From SUPERCOP
polarity of O-H bond. And hence the acid strength decreases.
b) Phenol is more acidic than alcohol: In phenol, the hydroxyl group is directly attached to
the sp2hybridised carbon of benzene ring which acts as an electron withdrawing group
whereas in alcohols, the hydroxyl group is attached to the alkyl group which have electron
releasing inductive effect. In phenol, the hydroxyl group is directly attached to the
sp2hybridised carbon of benzene ring whereas in alcohols, the hydroxyl group is attached to
the sp3hybridised carbon of the alkyl group. The sp2hybridised carbon has higher
electronegativity than sp3hybridised carbon. Thus, the polarity of O-H bond of phenols is
higher than those of alcohols. Hence, the ionisation of phenols is higher than that of alcohols.
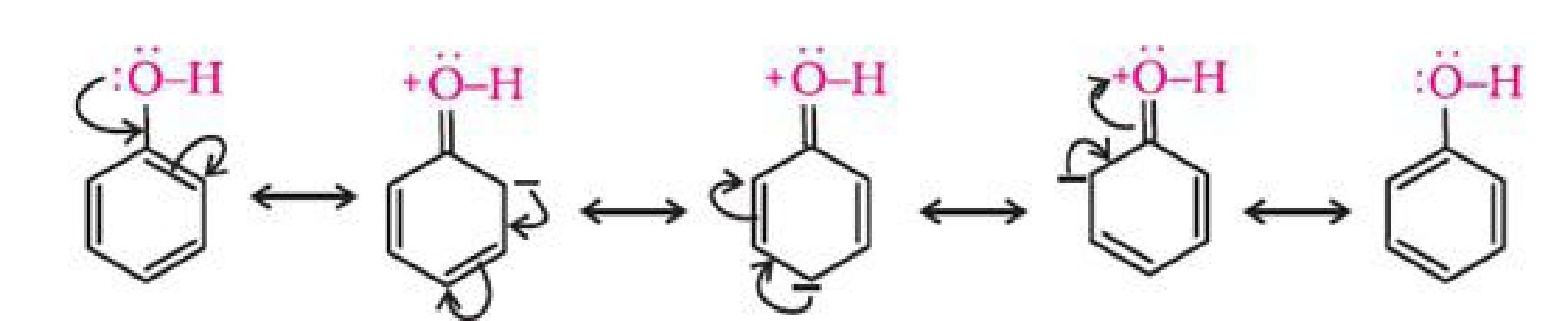
The ionisation of an alcohol and a phenol takes place as follows:
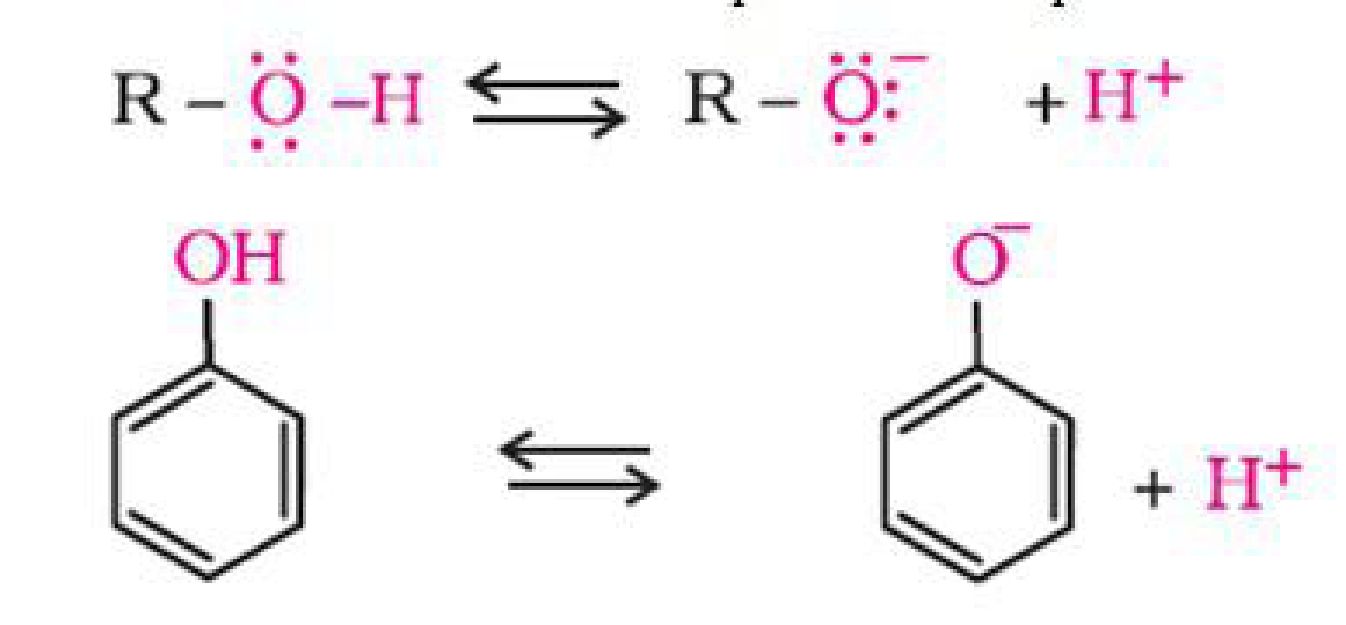
In alkoxide ion, the negative charge is localised on oxygen while in phenoxide ion, the charge
is delocalised.
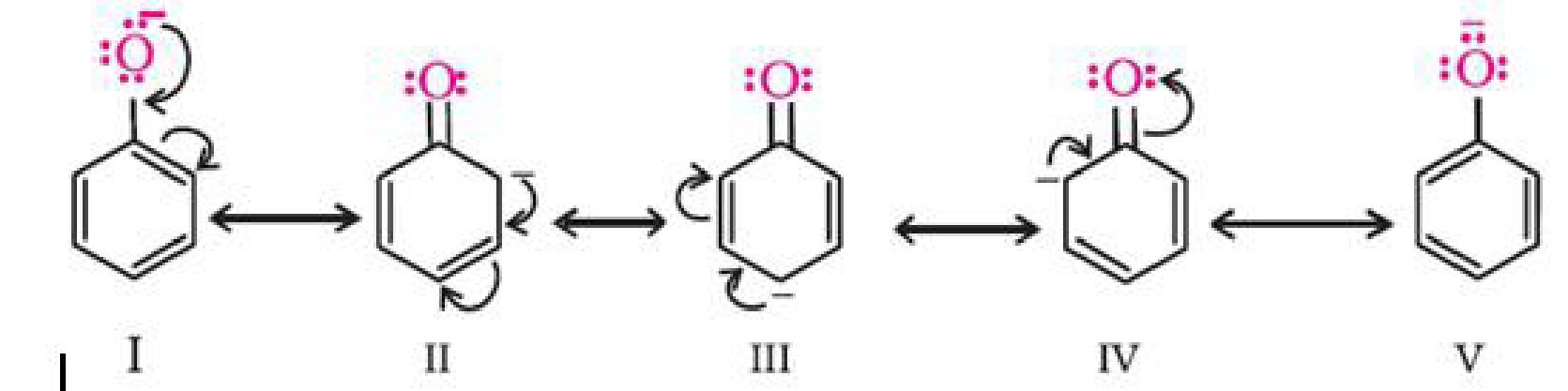
The delocalisation of negative charge makes phenoxide ion more stable and favours the
ionisation of phenol. Although there is also charge delocalisation in phenol, its resonance
structures have charge separation due to which the phenol molecule is less stable than
phenoxide ion.
- In substituted phenols, the presence of electron withdrawing groups such as nitro group
enhances the acidic strength of phenol. On the other hand, electron releasing groups, such as
alkyl groups, in general, decreases the acid strength. It is because electron withdrawing
groups lead to effective delocalisation of negative charge in phenoxide ion.
• Differentiate between organic compounds:
- Alcohols and phenols
Phenol on reaction with neutral FeCl3 gives purple colour whereas alcohols do not give
purple colour.
QCiH5OH + Fe3+ ^ [Fe(OCiH5)6}3– + 6
Purple colour
- Primary, secondary and tertiary alcohols
Lucas reagent test:
c0nc.HCl+ZnCl2/Lucas reagent
If it is a primary alcohol, no turbidity appears at room temperature. Turbidity appears only
on heating. If it is a secondary alcohol, turbidity appears in 5 minutes. If it is a tertiary
alcohol, turbidity appears immediately.
- Methanol and ethanol
Iodoform test: Ethanol when reacted with (I2 and NaOH) or NaOI gives yellow ppt of
iodoform since it has the presence of CH3-CH (OH)- group.
C2H5OH + 4J2 + WaOH^ CHI3 + hNaI + §H20 + HC00Na
Yellow ppt
CH3OH +12 + NaOH ^ No yellow ppt
sp^hytmdffied
i
Msthoatymfihajif
0fttec)
• Preparation of ethers:
a) From alcohols
H2SO4 or H3PO4 at413K
Alcohol > Ethers
b) From alkyl halide and sodium alkoxide
Williamson! s synthesis
Ethers < Alkyl halide and sodium alkoxide
Here, the alkyl halide should be primary and alkoxide should be tertiary. In case of aromatic
ether, the aromatic part should be with phenoxide ion.
• Physical properties of ethers:
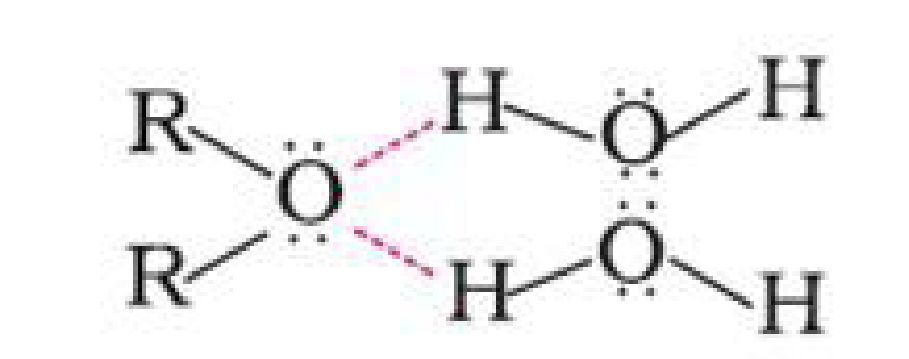
a) Miscibility: Miscibility of ethers with water resembles those of alcohols of the same
molecular mass. This is due to the fact that just like alcohols, oxygen of ether can also form
hydrogen bonds with water molecule.
Ethers have much lower boiling points than alcohols. This is due to the presence of hydrogen
bonding in alcohols. Hydrogen bonding is absent in ethers.
R R
1. J
l i
j H *
Chemical properties of ethers:
a) Cleavage of C-O bond in ethers:
R-O-R’ + HX ^ R-X + R’OH
Excess
The order of reactivity of hydrogen halides is as follows: HI >HBr>HCl
Alkyl halide formed is always the lower alkyl group. But if a tertiary alkyl group is present,
the alkyl halide is always tertiary. In case of phenolic ethers, the cleavage occurs with the
formation of phenol and alkyl halide.
b) Electrophilic substitution reaction in aromatic ethers:
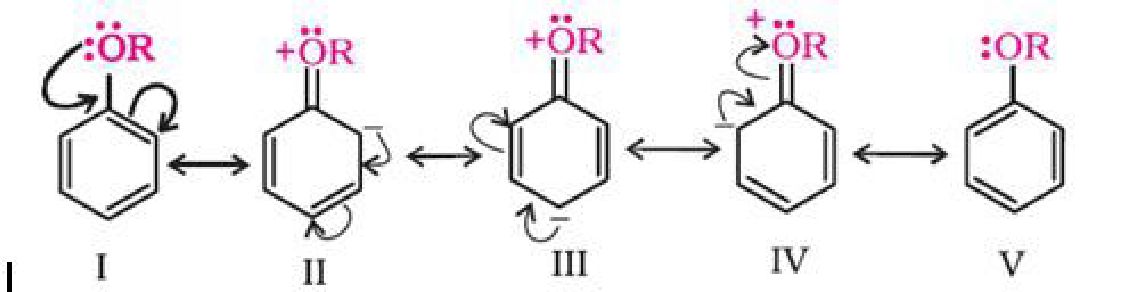
The electrophilic substitution reaction of aromatic ether involves the following reaction:
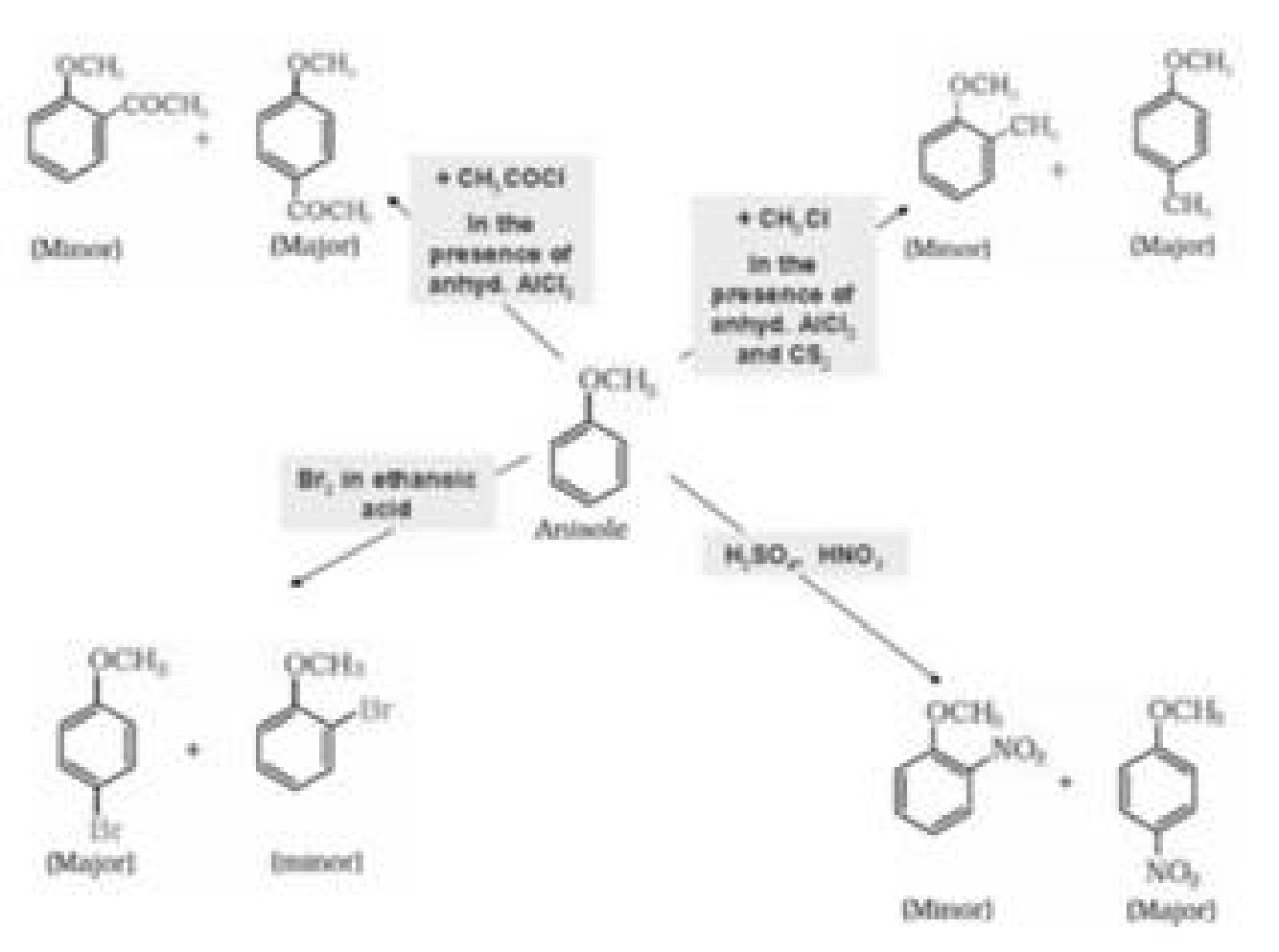
• Other conversion reactions:
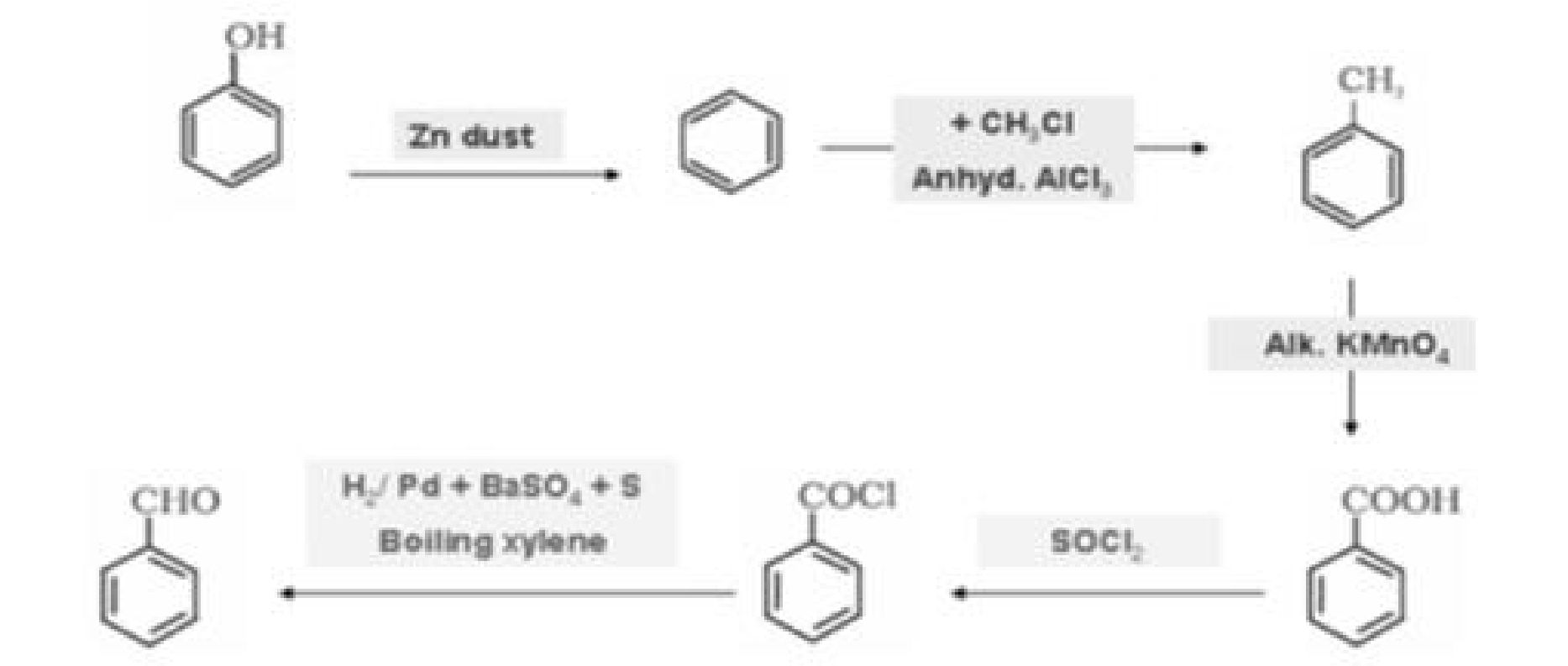
a) Phenol to salicyldehyde
b) Phenol to benzene diazonium chloride