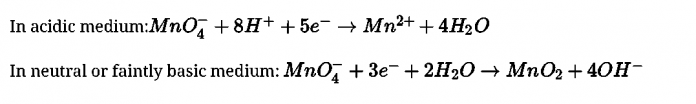CBSE Class 12 Chemistry
Quick Revision Notes
Chapter 8
- The elements lying in the middle of periodic table belonging to groups 3 to 12 are known as d – block elements.
- Their general electronic configuration is (n — 1) d1_10ns1_2 where (n – 1) stands for penultimate (last but one) shell.
- A transition element is defined as the one which has incompletely filled d orbitals in its ground state or in any one of its oxidation states.
- Zinc, cadmium, mercury are not regarded as transition metals due to completely filled d – orbital.
- The f-Block elements: The elements constituting the/-block are those in which the 4 fand 5forbitals are progressively filled in the latter two long periods.
- Lanthanoids: The 14 elements immediately following lanthanum, i.e., Cerium (58) to Lutetium (71) are called lanthanoids. They belong to first inner transition series. Lanthanum (57) has similar properties. Therefore, it is studied along with lanthanoids.
- Actinoids: The 14 elements immediately following actinium (89), with atomic numbers 90 (Thorium) to 103 (Lawrencium) are called actinoids. They belong to second inner transition series. Actinium (89) has similar properties. Therefore, it is studied along with actinoids.
- Four transition series:
- 3d – transition series. The transition elements with atomic number 21(Sc) to 30(Zn) and having incomplete 3d orbitals is called the first transition series.
- 4d – transition series. It consists of elements with atomic number 39(Y) to 48 (Cd) and having incomplete 4d orbitals. It is called second transition series.
- 5d – transition series. It consists of elements with atomic number 57(La), 72(Hf) to 80(Hg) having incomplete 5d orbitals. It is called third transition series.
- 6d – transition series. It consists of elements with atomic number 89(Ac), 104(Rf) to 112(Uub) having incomplete 6d orbitals. It is called fourth transition series.
• General Characteristics of transition elements:
- Metallic character: All transition elements are metallic in nature, i.e. they have strong metallic bonds. This is because of presence of unpaired electrons. This gives rise to properties like high density, high enthalpies of atomization, and high melting and boiling points.
- Atomic radii: The atomic radii decrease from Sc to Cr because the effective nuclear charge increases. The atomic size of Fe, Co, Ni is almost same because the attraction due to increase in nuclear charge is cancelled by the repulsion because of increase in shielding effect. Cu and Zn have bigger size because the shielding effect increases and electron electron repulsions repulsion increases.
- Lanthanoid Contraction: The steady decrease in the atomic and ionic radii of the transition metals as the atomic number increases. This is because of filling of 4f orbitals before the 5d orbitals. This contraction is size is quite regular. This is called lanthanoid contraction. It is because of lanthanoid contraction that the atomic radii of the second row of transition elements are almost similar to those of the third row of transition elements.
- Ionisation enthalpy: There is slight and irregular variation in ionization energies of transition metals due to irregular variation of atomic size. The I.E. of 5d transition series is higher than 3d and 4d transition series because of Lanthanoid Contraction.
- Oxidation state: Transition metals show variable oxidation states due to tendency of (n- 1)d as well as ns electrons to take part in bond formation.
- Magnetic properties: Most of transition metals are paramagnetic in nature as a result of which they give coloured compounds and it is all due to presence of unpaired electrons. It increase s from Sc to Cr and then decreases because number of unpaired and then decrease because number of unpaired electrons increases from Sc to Cr and then decreases. They are rarely diamagnetic.
- Catalytic properties: Most of transition metals are used as catalyst because of (i) presence
of incomplete or empty d – orbitals, (ii) large surface area, (iii) varuable oxidation state, (iv) ability to form complexes, e.g., Fe, Ni, V2O3, Pt, Mo, Co and used as catalyst.
- Formation of coloured compounds: They form coloured ions due to presence of incompletely filled d – orbitals and unpaired electrons, they can undergo d – d transition by absorbing colour from visible region and radiating complementary colour.
- Formation of complexes: Transition metals form complexes due to (i) presence of vacant d – orbitals of suitable energy (ii) smaller size (iii) higher charge on cations.
- Interstitial compounds: Transition metals have voids or interstitials in which C, H, N, B etc. can fit into resulting in formation of interstitial compounds. They are non – stoichiometric, i.e., their composition is not fixed, e.g., steel. They are harder and less malleable and ductile.
- Alloys formation: They form alloys due to similar ionic size. Metals can replace each other in crystal lattice, e.g., brass, bronze, steel etc.

- Potassium permanganate is prepared by fusion of MnO4 with alkali metal hydroxide (KOH) in presence of O2 or oxidising agent like KNO3. It produces dark green K2MnO4 which undergoes oxidation as well as reduction in neutral or acidic solution to give permanganate.
2MnO2 + 4KOH + O2 ^ 2K2MnOA + 2H2O 4H+ + 3MnO\~ ^ 2MnO^ + MnO2 + 2H2O
- Commercially, it is prepared by the alkaline oxidative fusion of MnO2 followed by the electrolytic oxidation of manganate (Vl).

- In laboratory, Mn2+ salt can be oxidized by peroxodisulphate ion to permanganate ion.
- +3 oxidation state is most common along with +2 and +4.
- Except Promethium, they are non – radioactive.
- The magnetic properties of lanthanoids are less complex than actinoids.
- Actinoids also show higher oxidation states such as +4, +5, +6 and +7.
- They are radioactive.
- The magnetic properties of the actinoids are more complex than those of the lanthanoids.
- They are more reactive.
- It is a well-known alloy which consists of a lanthanoid metal (~ 95%)and iron (~ 5%) and traces of S, C, Ca and Al.
- A good deal of mischmetall is used in Mg-based alloy to produce bullets, shell and lighter flint.
/ 4
Material Downloaded From SUPERCOP