Science for class-IX (NCERT)
Chapter 2: Is Matter Around Us Pure
2.1- Introduction
In the previous chapter, we learned about matter. In this chapter, we would discuss about mixtures, compounds, and elements. We would learn about different types of mixtures and discuss what matter is pure or not pure.
2.2- Mixture
When two or more pure forms of matter combine together, it is known as a mixture. These pure forms of matter are also known as substance.
Substance:-Any form of matter that cannot be separated into other kinds of matter by using any physical means is known as substance. For e.g.- sodium chloride or salt is a substance and cannot be separated into other forms by any means. Another example is water or H2O.
Some examples of mixture can be:
- Soft Drinks that are prepared by mixing various ingredients, flavors, and colors together.
- Cement is a mixture of limestone, silica and some other material.
- Air around us is itself a mixture of various gases like Oxygen, Nitrogen, Carbon Dioxide etc.
2.3- Types of mixture
Depending upon the nature of the constituents of the mixture, a mixture can be categorized into three types:
2.3.1- Solution
The homogenous mixture of two or more substances is called a solution. In a solution, all the particles of the constituents are evenly distributed within the mixture. The components of a solution are the solvent and the solute. Some examples of solutions are:
- Alloys, which are the mixtures of two or more metals or a metal and a non-metal that cannot be separated into their components by physical methods. Brass is an alloy containing 30% zinc and 70% copper.
- As discussed above, air is also a homogenous mixture of various gases that are uniformly mixed together in fixed percentage.
Also we have used words solvent and solute above, which we can define as follows:
Solvent:- The component of the solution that dissolves the other component in it is called solvent. Generally, it is the component present in a larger amount.
Solute:– The component of the solution that dissolves within the solvent is called solute. Generally, it is the substance present in a smaller amount.
Keeping above points in mind, we can find some more examples regarding a solution:
- A mixture of sugar in the water is an example of solid mixed inside a liquid solution. In this solution, water acts like the solvent and sugar act like solute.
- ‘Tincture of iodine’, which is a solution of iodine in alcohol, contains iodine as the solute and alcohol as the solvent.
- The aerated drinks like soda water, cola drinks, are solutions of gas inside liquid. They contain carbon dioxide as the solute and water as solvent.
- The air around us is a solution of gas inside gas. It contains gases like 21% Oxygen and 78% Nitrogen as the main components and numerous other gases that are present in a very small percentage.
Properties of a solution:-
- A solution is a homogeneous mixture.
- The particles of a solution are smaller than 1 nm (10-9 metre) and cannot be seen with naked eyes.
- Because of such small particles size, they do not scatter a beam of light passing through the solution. So, the path of light is not visible in a solution.
- The solute particles can’t be separated from the mixture using filtration. The solute particles do not settle down in a solution, so a solution is stable.
Concentration of a solution:-
Concentration refers to the amount of solute present in the solution in a given solution mixture. It is the amount of solute present per unit volume or per unit mass of the solution/solvent. There are three types of concentrations for a solution:
- Dilute solution: When the amount of solute present in the solution/solvent is low, then it is a dilute solution.
- Concentrated solution: When the amount of solute present in the solution/solvent is high, then it is a concentrated solution.
- Saturated solution: When the amount of solute present in the solution/solvent is equal to the full capacity (i.e. the solution has dissolved as much solute as it can hold) of the solution, then it is called a saturated solution. Also, if the amount of solute contained in a solution is less than the saturation level, it is called an unsaturated solution.
Solubility:-The maximum amount of solute which a solution can hold at the given temperature, or in other words, the maximum capacity of a solution for holding the solute particles at a given temperature is called the solubility of that solution.
Concentration of a solution=Amount of solute/Amount of solution
Or
Amount of solute/Amount of solvent
2.3.2- Suspension
A suspension is a non-homogeneous mixture in which the solid particles are dispersed in the liquid. In other words, a suspension is a heterogeneous mixture in which the solute particles do not dissolve but remain suspended throughout the bulk of the medium. The particles of a suspension can be seen with naked eyes. Some examples of a suspension are given below:
- A mixture of sand in water. The sand particles are insoluble and remain suspended.
- Muddy water is also a suspension of mud in water.
Properties of a suspension:-
- A suspension is a heterogeneous mixture.
- Particles of a suspension are visible to the naked eyes.
- Particles of a suspension can scatter a beam of light passing through them and make its path visible.
- Solute particles can settle down when left undisturbed and then, can be separated by the process of filtration. In other words, a suspension is an unstable mixture.
Note:When the particles of a suspension settle down, the suspension breaks and it does not scatter light anymore. This explains more clearly that why we call a suspension unstable.
2.3.3- Colloid or Colloidal solution
A colloidal solution or colloid is a heterogeneous mixture, in which the particles are uniformly spread in the solution. The particles are too small to be seen with naked eyes, but large enough to scatter a beam of light. Colloids are used in industries and also in daily life. There are two components of a colloid:
- Dispersed phase:The particles that are uniformly spread throughout the solution are called the dispersed phase.
- Dispersion medium: The medium in which the particles are spread is called the dispersion medium.
Before we talk more about colloidal solutions, let’s discuss about an important scientific phenomenon which we have been mentioning above:
Tyndall effect:-It is the phenomenon in which a beam of light gets scattered or in other words, the scattering of a beam of light is called the Tyndall effect. It was named after the scientist who had discovered it.
So, when we say that a suspension or colloidal solution can scatter a beam of light, we mean that a suspension or colloidal solution shows Tyndall effect.
This effect can also be observed when a small beam of light enters a room through a small hole. This is due to the scattering of light by the particles of dust and smoke in the air. It can also be observed when sunlight passes through the canopy of a dense forest. This is due to the mist, containing tiny droplets of water, which act as particles of colloid dispersed in air.
Properties of a colloid:-
- A colloid is a heterogeneous mixture.
- Particles of a colloid are too small to be seen with naked eyes.
- Particles of colloid are big enough to scatter a beam of light passing through it and make its path visible.
- Particles of a colloid do not settle down when left undisturbed, or in other words, a colloid is quite stable.
- Particles of a colloid cannot be separated from the mixture by filtration. However, a special technique called centrifugationcan be used to separate them.
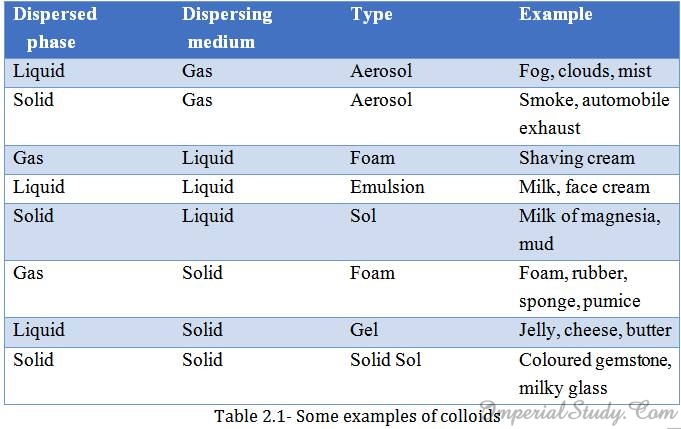
Note:Colloids are classified according to the state i.e. solid, liquid and gas, of the dispersing medium and the dispersed phase. In the following table given below, you can see some examples of colloids that we come across in our daily life:
2.4- Techniques used to separate the components of a mixture
The particles of heterogeneous mixtures can be easily separated by easy methods like sieving, handpicking and filtration. However, apart from these simple techniques, there are some special techniques that can be used to separate the components of mixture. In this section, we will discuss about some of these techniques with examples.
2.4.1- Evaporation
Evaporation is a separation technique used to separate a volatile component from its non-volatile solute. This method can be used to separate coloured component (dye) from blue/black ink. Ink is a mixture of dye in water. We can observe this technique with the help of the following activity:
- Take a beaker and fill it half with water.
- Now put a watch glass on the mouth of the beaker.
- Put a few drops of ink on the watch glass.
- Start heating the beaker. Observe the evaporation taking place from the watch glass.
- Keep heating until any further change cannot be observed
- You will see that the Dye has been separated from the blue/black ink.
2.4.2- Centrifugation
We have already talked about centrifugation earlier. Now we must discuss what centrifugation is and where it is used. This technique works on the principle that the denser particles are forced to the bottom and lighter particles stay at the top when spun rapidly. For this purpose, a centrifugation machine is used. (While sometimes a milk churner can also be used when the technique is used on full cream, toned or double-toned milk) Here is a small activity to understand centrifugation:
- Take some full-cream milk in a test tube.
- Use a centrifuging machine (or a milk churner) for two minutes.
- You can see that the cream has been separated from the milk.
Applications/Uses of Centrifugation:-
- It is used in the diagnostic laboratories for carrying out blood and urine tests.
- It is commonly used in dairies or at home to separate butter/cream from milk.
- It is used in washing machines to squeeze out the water from dirty clothes.
2.4.3- Separation of immiscible liquids
Immiscible liquids are liquids which cannot be mixed together. The technique used to separate two immiscible liquids is quite simple and we can observe it with the help of a small activity in which we try to separate kerosene oil from water using a separating funnel:
- Take the mixture of kerosene oil and water.
- Pour the mixture in a separating funnel.
- Let it undisturbed for some time until separate layers of oil and water are formed.
- Open the stopcock of the separating funnel and pour out the lower layer of water carefully.
- Now, close the stopcock as the oil reaches it.
- Kerosene oil and water have been separated.
Applications/Uses of using a separating funnel (or a similar tool):-
- This technique is used to separate the mixture of oil and water.
- This technique is used in the extraction of iron from its ore. For this purpose, the lighter slag is removed from the top to leave the molten iron at the bottom in the furnace.
This technique works on the principle that immiscible liquids separate out in layers depending on their densities.
2.4.4- Sublimation
Sublimation is a technique used to separate mixtures that contain a sublimable volatile component from a non-sublimable impurity. Please note that in the first chapter we have learnt that sublimation is the process of changing of state directly from solid into a gas. This process is thus also used in separation techniques. Some solids that sublime are ammonium chloride, camphor, naphthalene and anthracene. Below is a small activity to show separation of ammonium chloride and salt mixture by sublimation:
- Take a china dish and put some mixture of ammonium chloride and salt on it.
- Take a funnel and put it inverted over the china dish.
- Now put a cotton plug in the mouth of the inverted funnel.
- Start heating the china dish. Vapours of ammonium chloride would be formed inside the apparatus and solidified ammonium chloride would be deposited on the walls of the funnel.
- Keep heating until there are no more vapours.
- Remove the funnel. You would find that the salt is left behind on the china dish.
2.4.5- Chromatography
The name of this technique has been derived from the word ‘Kroma’ of Greek language which means ‘Colour’. This is due to the fact that this technique was first used in the separation of colours.
Chromatography is the technique that is used to separate those solutes which dissolve in the same solvent. Below is an activity to show how to separate different colours from black ink using chromatography:
- Take a thin strip of filter paper.
- Take a pencil and draw a line on the filter paper. This line should be approximately 3 cm above the lower edge.
- At the centre of the line, put a small drop of ink from a sketch pen or fountain pen. Let this ink dry.
- Take a test tube (or a glass, beaker, jar, etc) containing water and lower the filter paper into it in such a way that the drop of ink is just above the water level. Leave it undisturbed.
- As the water rises up, you would observe different colours on the filter paper strip i.e. different colours have been separated from black ink.
Note:In the above activity, the ink has water as the solvent and the black ink/dye as the solute. As the water level rises, the filter paper takes along the dye particles with it. A dye is generally a mixture of two or more colours. So, the colour that is more soluble in water rises faster and the colours get separated from the black ink/dye.
Applications/Uses of Chromatography:-
- It is used to separate colours from a dye.
- It is used to separate pigments from natural colours.
- It is used to separate drugs from blood.
2.4.6-Distillation
In the above sections, we have discussed about separating two immiscible liquids. In this section we will discuss about a special technique called distillation that is used to separate two miscible liquids.
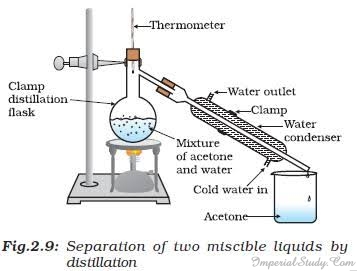
Distillation is used for the separation of components of a mixture containing two miscible liquids which boil without decomposition and have sufficient difference in their boiling points. Below is a small activity that shows how to separate a mixture of acetone and water with the help of distillation:
- Take the mixture of acetone and water.
- Pour it in a distillation flask and fit it with a thermometer.
- Attach the flask with a clamp and a water condenser on one side.
- Put a jar/beaker at the outlet of the water condenser.
- Heat the mixture slowly, closely watching the thermometer.
- Observe that the acetone vaporizes, condenses in the condenser and can be collected from the outlet into the jar/beaker.
- Observe how the water is left behind in the distillation flask.
Fractional Distillation:-
We have discussed the distillation process above. However, there is another related process called fractional distillationwhich we would discuss in this same section.
Fractional distillation is the process used to separate a mixture of two or more miscible liquids for which the difference in their boiling points is less than 25 K. Some applications of this process are the separation of different gases from the airand separation of different factions from petroleum products. The apparatus used in this process is similar to that of a simple distillation, except for that a fractionating column is fitted in between the distillation flask and the condenser.
Fractionating column:It is a tube packed with glass beads. The glass beads provide surface area for the vapours to cool and condense repeatedly.
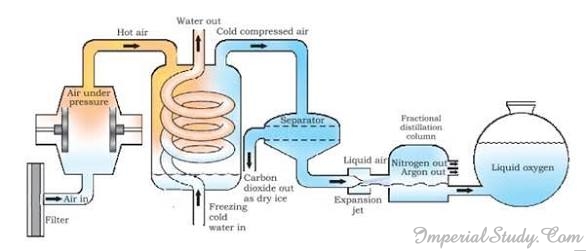
2.4.7- Separation of different components of air
We have already discussed before that the air is a mixture of various gases like oxygen, nitrogen, hydrogen, helium etc. Is there a way to separate these gases from the air? The answer is- fractional distillation. In the previous section, we mentioned fractional distillation. In this section, we will discuss that how this technique is used in the separation of various gases from the air around us.
Suppose we want a specific gas, say oxygen, out of the air mixture, then we simply need to separate out all the other gases present in the air. This process can be understood by the help of following steps:
- Firstly, the air is compressed by increasing the pressure.
- Then, it is cooled by decreasing the temperature and liquid air is obtained.
- The liquid air is allowed to warm up slowly in a fractional distillation column.
- The different gases in air get separated at different heights depending upon their boiling points.
The first gas that gets separated is Carbon dioxide that separates out as dry ice after the cold compressed air reaches the ‘separator’. Next gas that separates is nitrogen followed by argon that separate in the fractional distillation column. Oxygen in the form of liquid (liquid oxygen) is left behind and thus we separate oxygen from the mixture of air.
2.4.8- Crystallisation
The last technique that we will discuss in this section is crystallisation. Crystallisation is a process that separates a pure solid from a solution in the form of its crystals. This method is used to purify solids. For instance, the salt we obtain from seawater is not pure and can have lots of impurities within it. For such impurities, we use the crystallisation process. We can understand this process with a simple activity:
- Take about 5 g of impure sample of copper sulphate (CUSO4) in a china dish.
- Dissolve it in the least amount of water.
- Filter out the impurities using filtration.
- Now evaporate the water from the copper sulphate solution so that you get a saturated solution.
- Now cover this solution with a filter paper.
- Leave it undisturbed for a day at room temperature so that it cools down.
- After a day, you will obtain the crystals of copper sulphate in the china dish.
Now another question that arises is that which process is better? The similar evaporation process or the crystallisation process? The answer is crystallisation. Why?
Why the crystallisation method is better than evaporation:-
Although there may be many reasons to elaborate the answer, below are two main reasons to answer this question that has arisen:
- Evaporation is not a better technique because some solids may get decomposed and even some solids like sugar may get charred/burned on heating to dryness.
- Evaporation is not a better technique because some impurities may remain dissolved in the solution even after filtration and on evaporating, these impurities may contaminate the solid.
Applications/Uses of Crystallisation:-
- Crystallisation is used in the purification of salt that we obtain from seawater.
- It is also used in the separation of crystals of alum (phitkari) from impure samples.
So, these are the few techniques that are used in the separation of the components of mixture depending upon the nature of the components of the mixture. Now, it is time to discuss about an important concept which have already studied in previous classes and see how this concept is useful to understand this chapter.
2.5- Physical and Chemical changes
So as we have discussed in the previous chapter, the concept is necessary to understand the difference between a pure substance and a mixture. Let us understand this concept with the help of few terms:
- Physical properties:The properties of a substance that can be observed and specified are called physical properties. For example- colour, hardness, rigidity, fluidity, density, melting point, boiling point, etc. are all physical properties.
- Physical change:A change in the physical property of any solid, liquid or gas is called a physical change. So, change in colour, change in shape, change in state are all physical changes.
The interconversion of states (or change of state) is also a physical change because it has no effect on the chemical composition or chemical nature of that substance.
- Chemical properties: The properties of a substance that determine its chemical nature and chemical composition, like how the substance would react with fire, how does the substance smells, what is the chemical composed of, etc. are all the chemical properties of that substance.
- Chemical change: A change in the chemical composition or chemical properties of a substance is called a chemical change. An important characteristic of chemical change is that new substances are formed in a chemical change. A chemical change is also called a chemical reaction. You would study more about chemical reactions in Class 10th,
Burning of candle:During the burning of a candle, both physical and chemical changes take place. The wax is melted, which is a physical change while the carbon dioxide gas that is evolved is a chemical change in the wax.
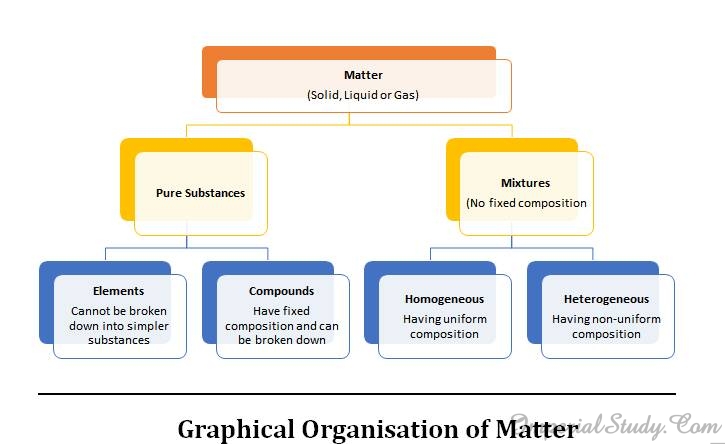
2.6-Types of Pure substances
Now when we have already discussed a lot about mixtures of different substances, it is finally the time to discuss about pure substances and their types. This concept is really necessary to understand as you would be learning more about them in upcoming chapters as well as in higher classes.
On the basis of their chemical composition, we can classify the substances as elements and compounds. Let us discuss about them in detail:
2.6.1- Elements
An element is a basic form of matter that cannot be broken down into simpler substances by using chemical reactions. This definition of an element has been given by a French chemist, Antoine Laurent Lavoisier (1743-94). Furthermore, Robert Boyle was the first scientistthat first used the term element in 1661. Elements are generally divided into metals, non-metals and metalloids.
Metals and their properties:-Metals are hard, shiny substances that generally show some or all of these properties:
- They have a lustre (shine).
- They have silvery-grey or golden-yellow colour.
- They conduct heat and electricity.
- They can be drawn into wires i.e. they are ductile.
- They can be hammered into thin sheets i.e. they are
- They make a ringing sound when they are hit i.e. they are
Some metals are iron, zinc, copper, gold, silver, sodium, potassium, etc. Mercury is the only metal that is liquid at room temperature.
Non –metals and their properties:- Non-metals are soft, non-conductors of heat and electricity that show some or all of these properties:
- They are available in a variety of colours.
- They are poor conductors of heat and electricity.
- They are neither lustrous, sonorous, malleable or ductile.
Some non-metals are hydrogen, iodine, carbon, bromine, chlorine, etc.
Metalloids: The elements that have intermediate properties between metals and non-metals, i.e. some properties of metals and some properties of non-metals are called metalloids. Some metalloids are boron, silicon and germanium.
Now coming back to elements, here are some important facts about elements that you must look at:
- There are more than 100 elements known today out of which ninety-two are naturally occurring while the rest are man-made.
- Most of the elements known at present are solid.
- Eleven elements are in gaseous state at room temperature.
- Two elements are liquid at room temperature i.e. mercury and bromine.
- The elements Gallium and Cesium become liquid at a temperature slightly above the room temperature. (303 K)
2.6.2- Compounds
A compound is a substance which is composed of two or more elements which are chemically combined with one another in a fixed proportion or a fixed amount. Examples of compounds are sulphur chloride, hydrogen sulphide, ammonium sulphate, and even water. You can easily conclude that in a compound, say sulphur chloride, the elements sulphur and chlorine are combined in fixed proportions
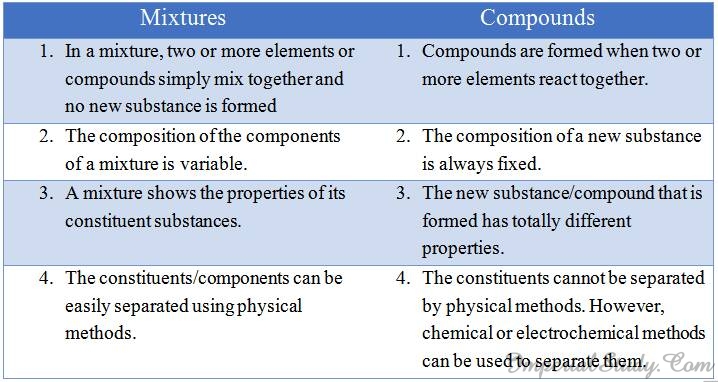
Difference between mixtures and compounds:-
Look at the following table in order to understand the difference between mixtures and compounds: